
It is a strong metal with low density that is quite ductile (especially in an oxygen- free environment),lustrous, and metallic-white in color. Metals Weight Calculator– Aluminum| Brass|Bronze| Copper|Magnesium|Plastic| Nickel| Stainless Steel| Steel| Titanium|ZincĪ metallic element, titanium is recognized for its high strength-to-weight ratio. Pipes Tubes Plates Bars Square Tubes Weight Calculation CalculatorĬonversion Calculator Calculation-Pressure|Weight|Temperature|Volume|LengthĬonversion Table-Pressure|Stress|Mass|Length|Temperature|SI Prelxes Titanium’s properties are chemically and physically similar to zirconium, because both of them have the same number of valence electrons and are in the same group in the periodic table. There are two allotropic forms and five naturally occurring isotopes of this element, 46Ti through 50Ti, with 48Ti being the most abundant (73.8%).
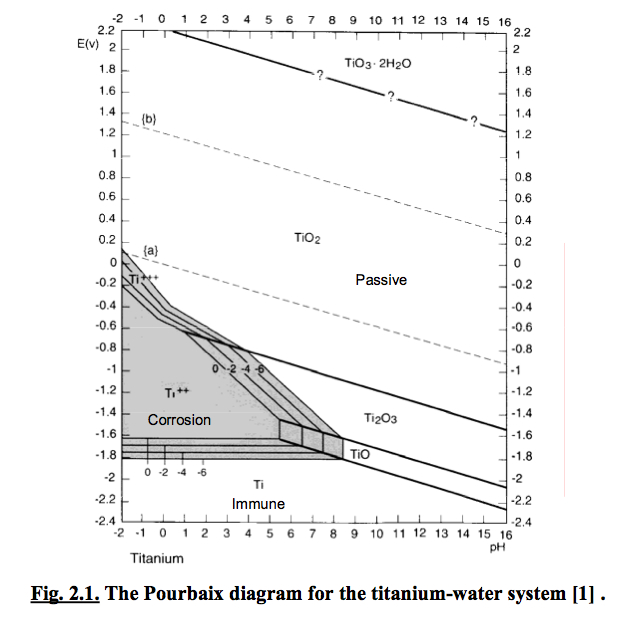
In its unalloyed condition, titanium is as strong as some steels, but 45% lighter. The two most useful properties of the metal form are corrosion resistance and the highest strength-to-weight ratio of any metal. Titanium can be alloyed with iron, aluminium, vanadium, molybdenum, among other elements, to produce strong lightweight alloys for aerospace (jet engines, missiles, and spacecraft), military, industrial process (chemicals and petro-chemicals, desalination plants, pulp, and paper), automotive, agri-food, medical prostheses, orthopedic implants, dental and endodontic instruments and files, dental implants, sporting goods, jewelry, mobile phones, and other applications. Other compounds include titanium tetrachloride (TiCl4), a component of smoke screens and catalysts and titanium trichloride (TiCl3), which is used as a catalyst in the production of polypropylene). Its most common compound, titanium dioxide, is a popular photocatalyst and is used in the manufacture of white pigments. The metal is extracted from its principal mineral ores via the Kroll process or the Hunter process. The element occurs within a number of mineral deposits, principally rutile and ilmenite, which are widely distributed in the Earth’s crust and lithosphere, and it is found in almost all living things, rocks, water bodies, and soils. Titanium was discovered in England by William Gregor in 1791 and named by Martin Heinrich Klaproth for the Titans of Greek mythology. Sometimes called the “space age metal”,it has a low density and is a strong, lustrous, corrosion-resistant (including seawater, aqua regia and chlorine) transition metal with a silver color. Titanium is a chemical element with the symbol Ti and atomic number 22. Although passive region has been included in the study, it was unclear from the literature if there was a passive film or how protective it was.Titanium | Titanium Alloy Grade Comparison Table | Lightweight Titanium Heat Exchangers for Airborne Applications | Titanium And Titanium-Base Alloys | Titanium Alloys Specifications | Titanium Alloys | Titanium Chemical Composition | Corrosion Resistance | Comparison Titanium | ASTM B265 | ASTM B265 Chemical Composition It was found that the stability of carbides is described only by the immune region of the Pourbaix diagram for carbides. It was found that the diagrams are able to explain the results of experimental work performed on chromium carbides in NaOH. Using the thermodynamic data available for the main species at 298 K (25 ✬), Pourbaix diagrams for the chromium carbides are constructed at a concentration of 10-6 M of aqueous species. Although passive region has been included in the study, it was unclear from the literature if there was a passive film or how protective it was.ĪB - The Pourbaix diagrams also known as the E-pH diagrams were constructed for hardfacing alloys based on three chromium carbides: Cr7C3, Cr23C6 and Cr3C2 at 298 K (25 ✬). N2 - The Pourbaix diagrams also known as the E-pH diagrams were constructed for hardfacing alloys based on three chromium carbides: Cr7C3, Cr23C6 and Cr3C2 at 298 K (25 ✬). T1 - Significance of Pourbaix Diagrams to Study the Corrosion Behaviour of Hardfacing Alloys Based on Chromium Carbides at 298 K (25 ☌)
